|
Good documentation practice (GDocP) is a crucial component of GMP compliance. Good Documentation Practices (GDocP) | GMP Basics GMP Basics of Good Documentation Practices (GDocP) Medicinal Cannabis Industry Conference Sponsors.International GMP Conference 2022 – Speakers.Medicinal Cannabis Cultivation & Regulations.Medicinal Cannabis Conference 2020 – 16/17 November.Cultivation Training Course & Dinner Update.Medicinal Cannabis Cultivation Training.Medicinal Cannabis Consulting Firms | Directory & Consulting.Applying for Licences to Manufacture Medicinal Cannabis API.Applying for Licences to Manufacture Medicinal Cannabis – FDF.Obtaining a Medicinal Cannabis Cultivation Licence in Australia.Extraction, Winterization & Filtration Stages. 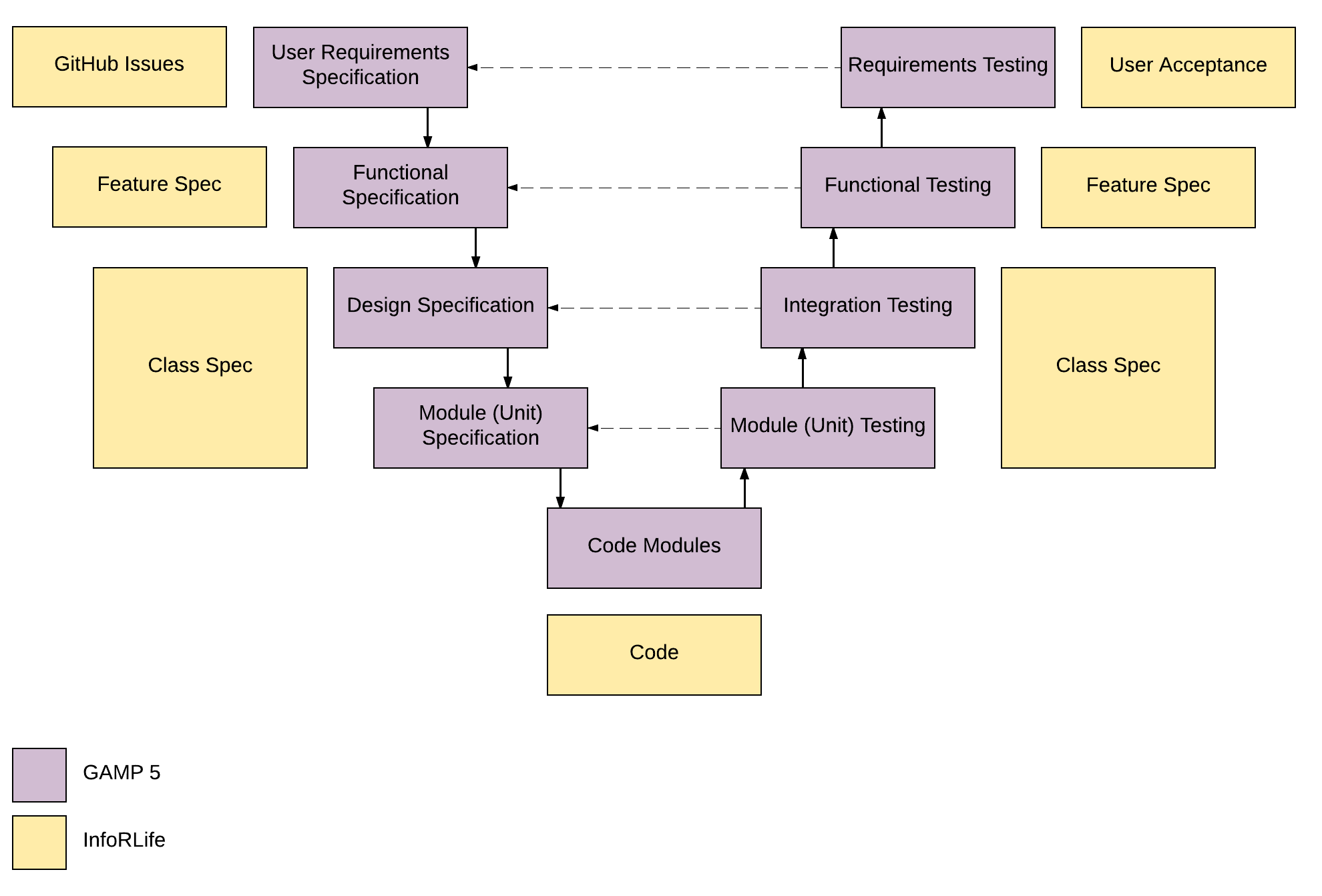 
Propagation, Germination (seeds) & Cloning Stages.Turn Key Medicinal Cannabis Design & Compliance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
